COVID-19 Vaccination Information
Upcoming COVID-19 Vaccination Opportunities
The ACRC/Safeway Pharmacy partnership, along with our service providers, to date, has led to over 9,000 of our clients being vaccinated as well as many family caregivers and service providers.
Get Vaccinated!
If you are in need of an in-home vaccination, please review this Update Letter.
The Client Advisory Committee came together virtually to produce a video on why they got the COVID-19 vaccine and why they hope you will get the vaccine, too!
Visit the CDC website for information on COVID Booster shots. California has a website available regarding vaccinations.
Pfizer Booster Shot
The Federal Centers for Disease Control and Prevention (CDC) issued updated temporary guidance for the people defined below who already were fully vaccinated with the Pfizer-BioNTech COVID-19 vaccine to receive a Pfizer-BioNTech COVID-19 booster shot to help increase their protection from COVID-19.
According to the announcement, the CDC recommends:
- people 65 years and older and residents in long-term care settings should receive a booster shot of Pfizer-BioNTech’s COVID-19 vaccine at least 6 months after their Pfizer-BioNTech primary series,
- people aged 50–64 years with underlying medical conditions should receive a booster shot of Pfizer-BioNTech’s COVID-19 vaccine at least 6 months after their Pfizer-BioNTech primary series,
- people aged 18–49 years with underlying medical conditions may receive a booster shot of Pfizer-BioNTech’s COVID-19 vaccine at least 6 months after their Pfizer-BioNTech primary series, based on their individual benefits and risks, and
- people aged 18-64 years who are at increased risk for COVID-19 exposure and transmission because of occupational or institutional setting may receive a booster shot of Pfizer-BioNTech’s COVID-19 vaccine at least 6 months after their Pfizer-BioNTech primary series, based on their individual benefits and risks.
Please note that “underlying medical conditions” above is defined in detail at those links, and includes Down Syndrome, cancer, diabetes, and immunocompromised states. The Governor issued a statement on September 24, 2021 about the Western States Scientific Safety Review Workgroup’s booster shot recommendations.
COVID-19 Vaccinations for Children
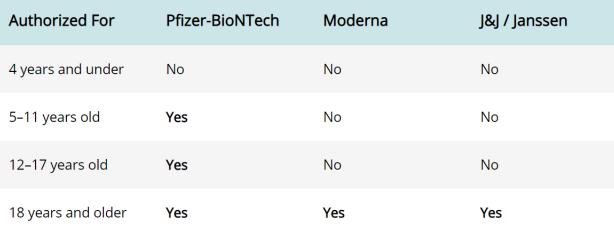
The Department of Developmental Services issued a directive describing the Federal Centers for Disease Control and Prevention (CDC) authorization and recommendations for the Pfizer-BioNTech COVID-19 vaccine to be administered to all children ages 5 through 11. The Western States Scientific Safety Review Workgroup also issued its confirmation. This news builds upon existing authorizations for children ages 12 and up. Within the announcement, the CDC provided the following helpful visual:
Starting April 15: COVID-19 Vaccination Eligibility Information
Anyone in California age 16 and over is now eligible to receive the vaccination.
Questions and Answers Regarding the COVID-19 Vaccine
From Ad Council: “It’s normal to be cautious when something new comes along. Wanting to know more is a good thing — it means you want to be informed. And getting informed about COVID-19 vaccines is an important step to help us stop this pandemic.” Visit their website to learn about the vaccine. You can also learn about other Ad Council campaigns here.
COVID-19 Vaccine Finder
Find a vaccination clinic near you by visiting vaccinefinder.org.
What to Expect After Getting the COVID-19 Vaccine
COVID-19 vaccination will help protect you from getting COVID-19. You may have some side effects, which are normal signs that your body is building protection. These side effects may feel like flu and may even affect your ability to do daily activities, but they should go away in a few days. Additional information regarding the vaccination. Información sobre la vacunación. CDC released plain language guides on COVID-19 vaccinations available for review.
Common Side Effects:
- Swelling and pain on the arm where you received the shot
- Fever
- Headache
- Chills
- Tiredness